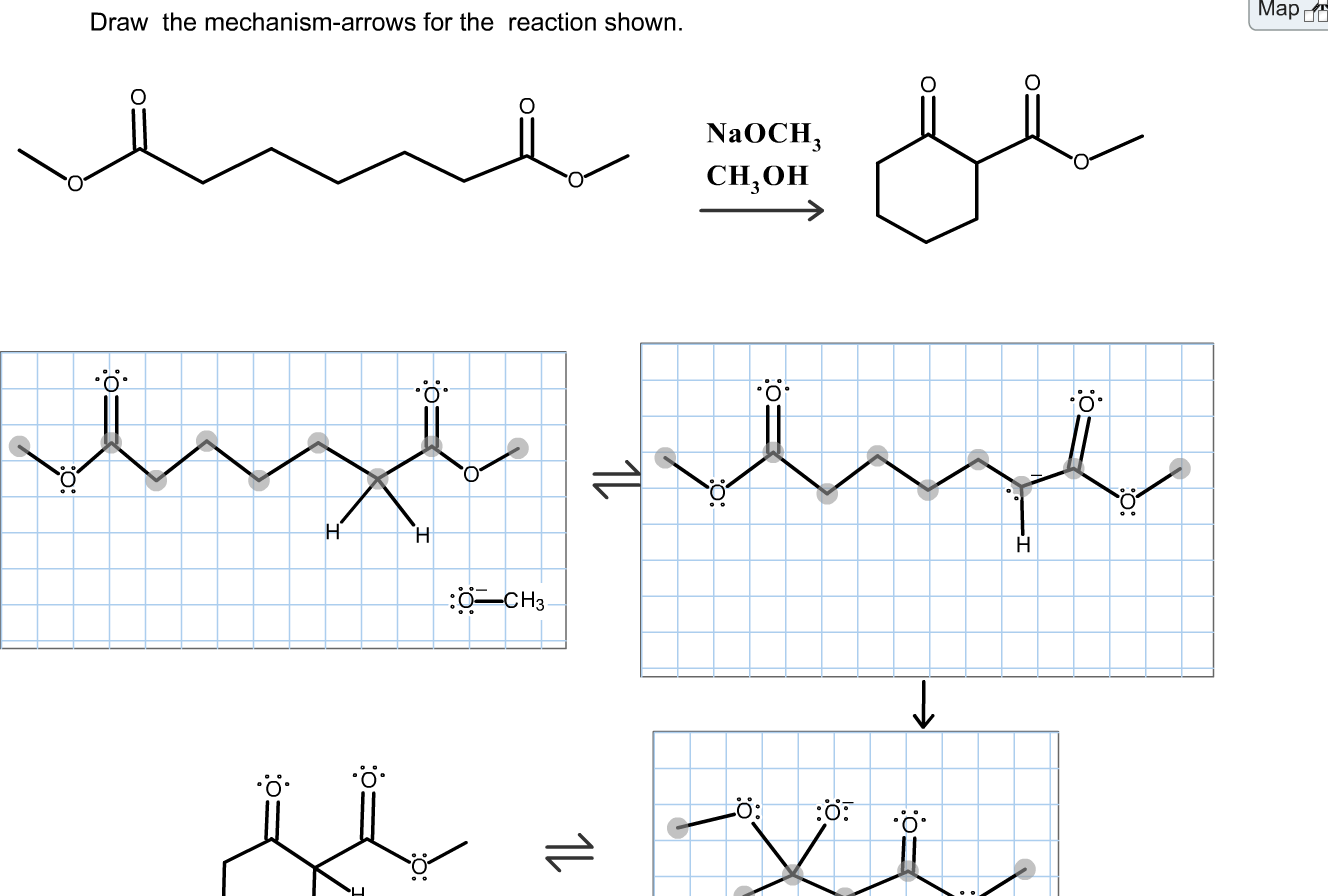
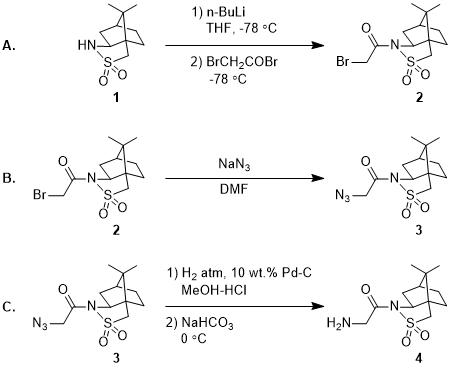
A Rapid Injection NMR Study of the Reaction of Organolithium Reagents with Esters, Amides, and Ketones - ScienceDirect

Are n-BuLi/TMEDA-Mediated Arene Ortholithiations Directed? Substituent-Dependent Rates, Substituent-Independent Mechanisms | Journal of the American Chemical Society

Metalation of Pyridines with nBuLi−Li−Aminoalkoxide Mixed Aggregates: The Origin of Chemoselectivity | Journal of the American Chemical Society

29 Si CPMAS NMR spectra of (a) carrier C, (b) C treated with BuLi, and... | Download Scientific Diagram

Enediolate–Dilithium Amide Mixed Aggregates in the Enantioselective Alkylation of Arylacetic Acids: Structural Studies and a Stereochemical Model | Journal of the American Chemical Society

Metalation of Pyridines with nBuLi−Li−Aminoalkoxide Mixed Aggregates: The Origin of Chemoselectivity | Journal of the American Chemical Society
![Lewis acid–catalyzed domino generation/[2,3]-sigmatropic rearrangement of ammonium ylides to access chiral azabicycles | Science Advances Lewis acid–catalyzed domino generation/[2,3]-sigmatropic rearrangement of ammonium ylides to access chiral azabicycles | Science Advances](https://www.science.org/cms/asset/860963e6-85a5-48b3-9e47-4a596c258578/abd5290-f1.gif)




![Stats] 2. Bundesliga table after matchday 25. : r/soccer Stats] 2. Bundesliga table after matchday 25. : r/soccer](https://preview.redd.it/7h0vyfk96xl81.jpg?auto=webp&s=ef20eb5c5e005905b30e2903b65f90e8ae894f6c)