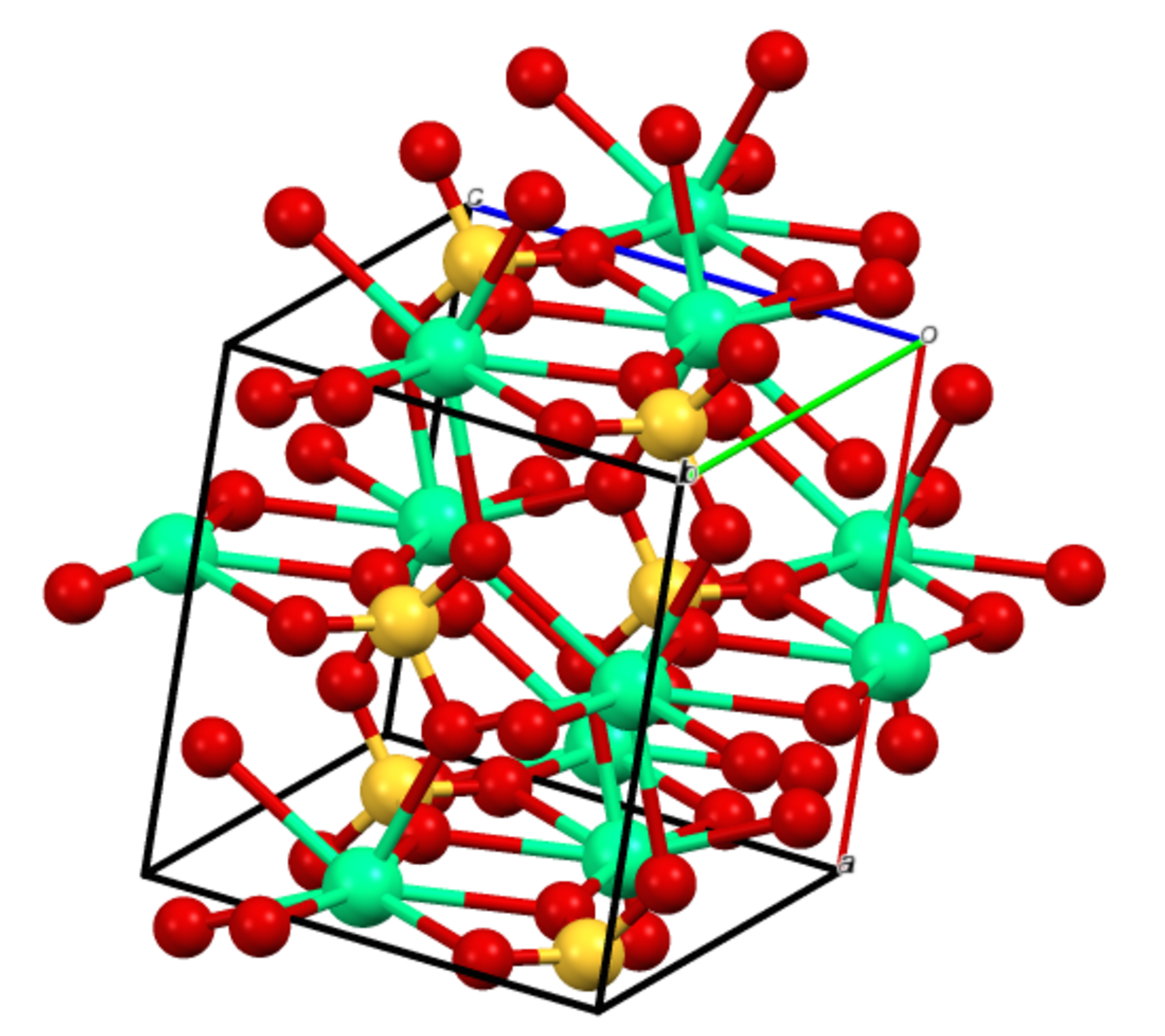
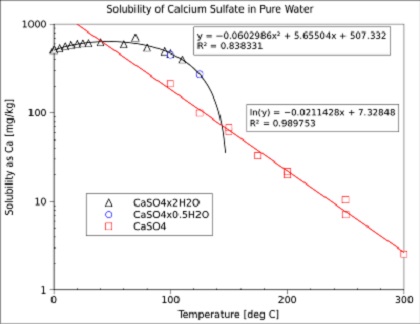
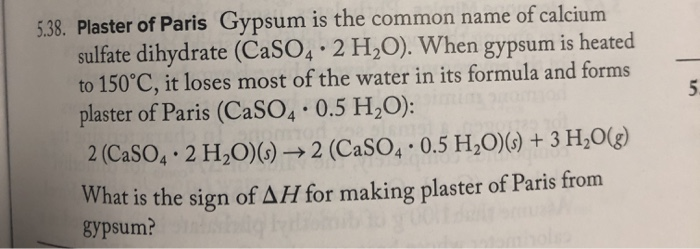Formation and Transformation of Five Different Phases in the CaSO4−H2O System: Crystal Structure of the Subhydrate β-CaSO4·0.5H2O and Soluble Anhydrite CaSO4 | Chemistry of Materials
Force field for calcium sulfate minerals to predict structural, hydration, and interfacial properties
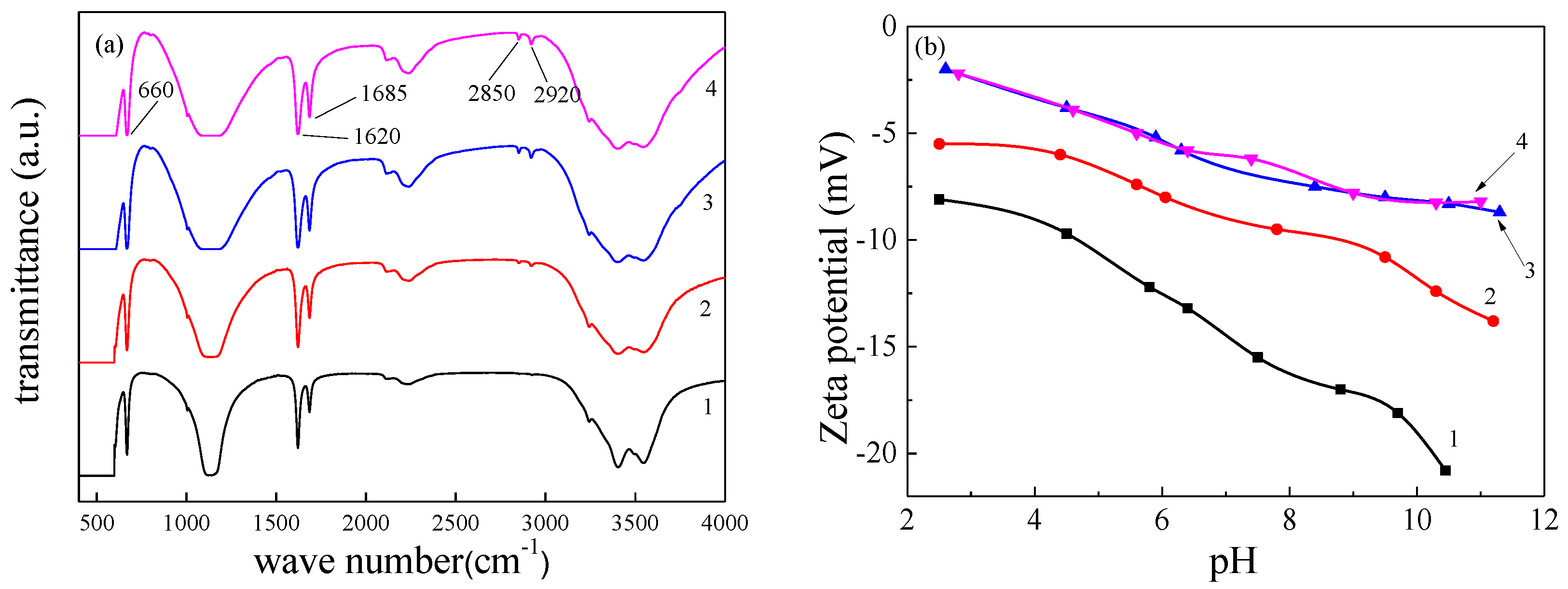
Crystals | Free Full-Text | Influence of Alkyl Trimethyl Ammonium Bromides on Hydrothermal Formation of α-CaSO4·0.5H2O Whiskers with High Aspect Ratios
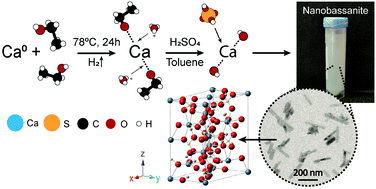
Synthesis of high surface area CaSO4·0.5H2O nanorods using calcium ethoxide as precursor - Chemical Communications (RSC Publishing)

The effect of additives on the hydration of CaSO4·0.5H2O: A synchrotron X-ray diffraction study - ScienceDirect
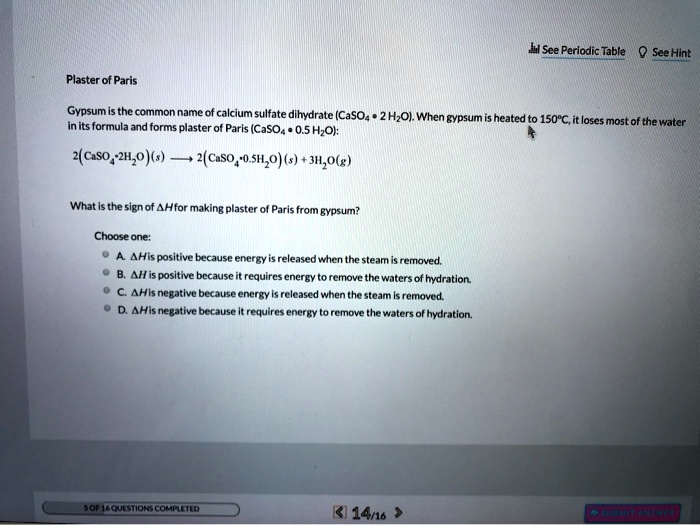
SOLVED: H See Perlodic Table Sce Hint Plaster of Paris Gypsum the common name calcium sulfate dihydrate (CaSOa- 2 HzOl. When gypsum heated 150*C,it loses [ most of the water inits formula

SOLVED: Gypsum is the common name of calcium sulfate dihydrate which has the formula CaSO4· 2 H2O When gypsum is heated to 150^∘C, it loses most of the water in its formula

Formation and Transformation of Five Different Phases in the CaSO4−H2O System: Crystal Structure of the Subhydrate β-CaSO4·0.5H2O and Soluble Anhydrite CaSO4 | Chemistry of Materials

Crystal structure of (a) γ‐CaSO4 and (b) bassanite CaSO4 ⋅ 0.5H2O, as... | Download Scientific Diagram