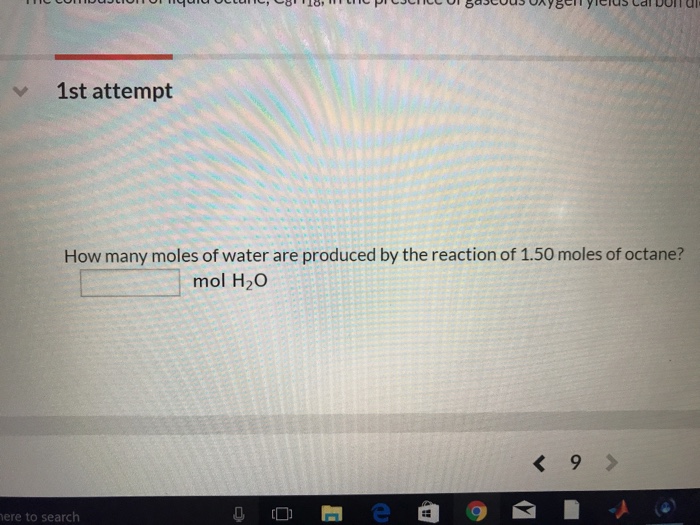
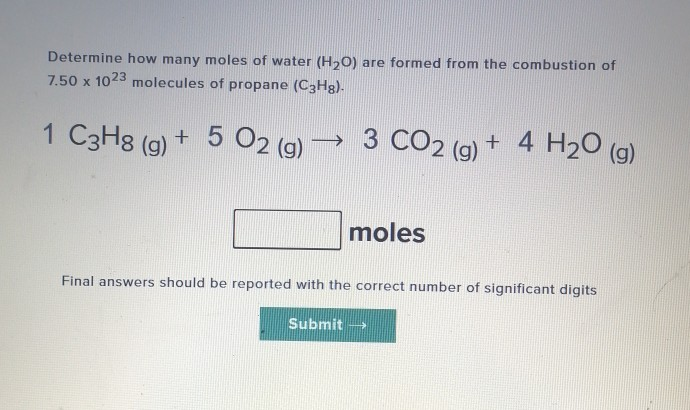
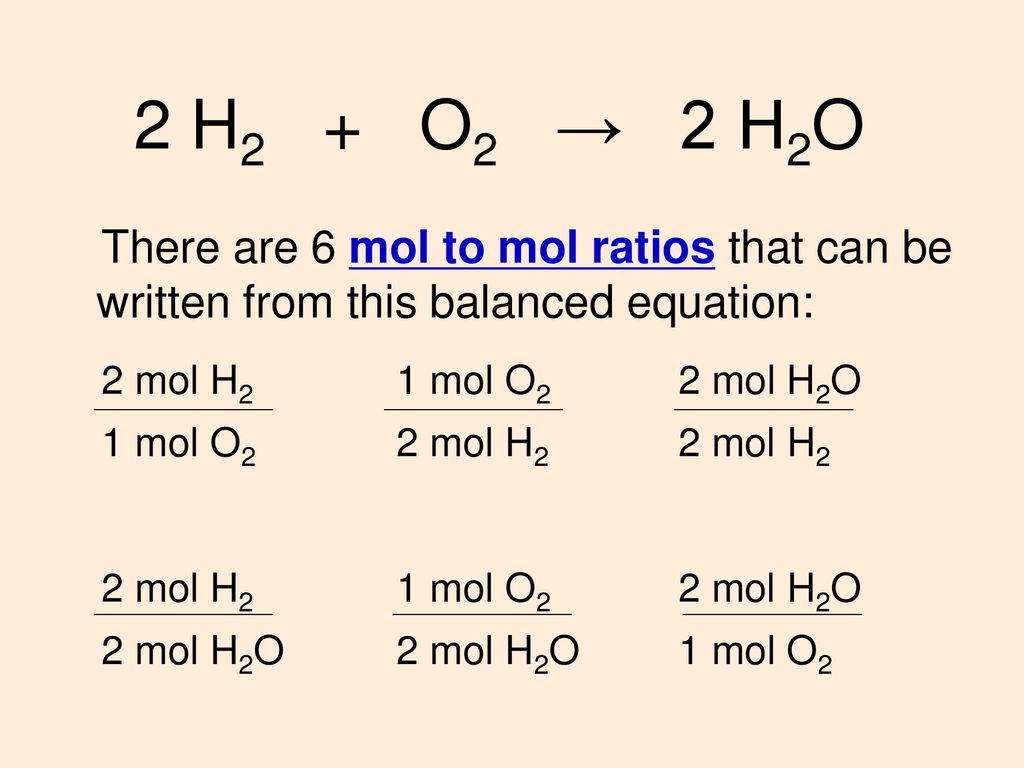
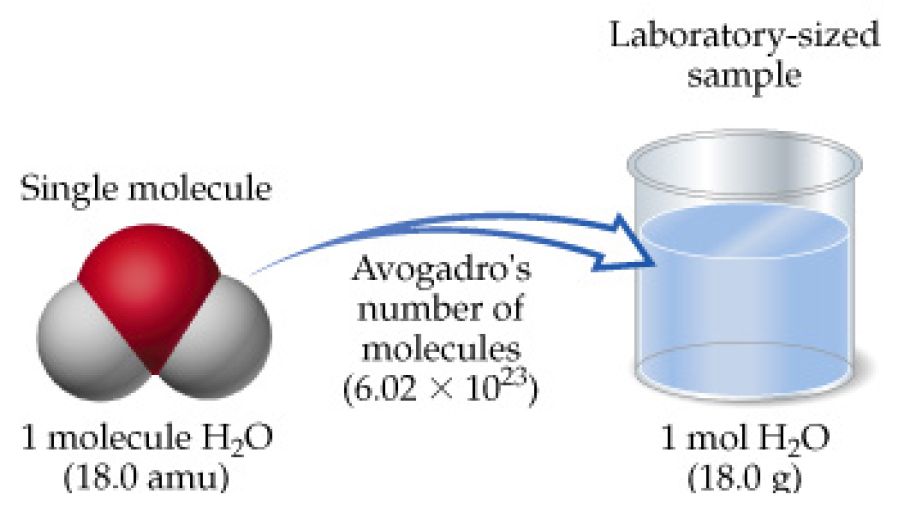
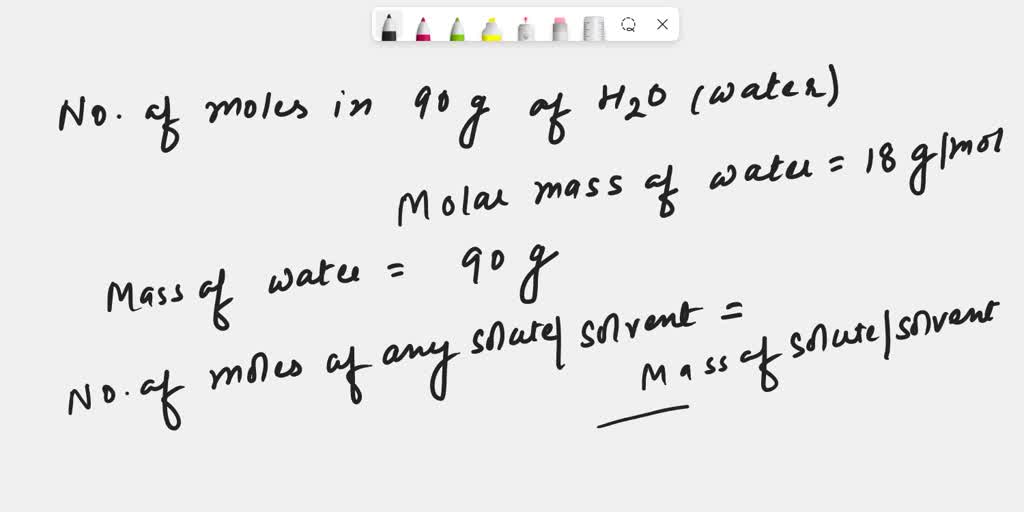SOLVED: How many moles of hydrogen atoms are present in a sample that contains 3.64 moles of water, H2O? answer=mol H How many moles of H2O molecules are present in a sample
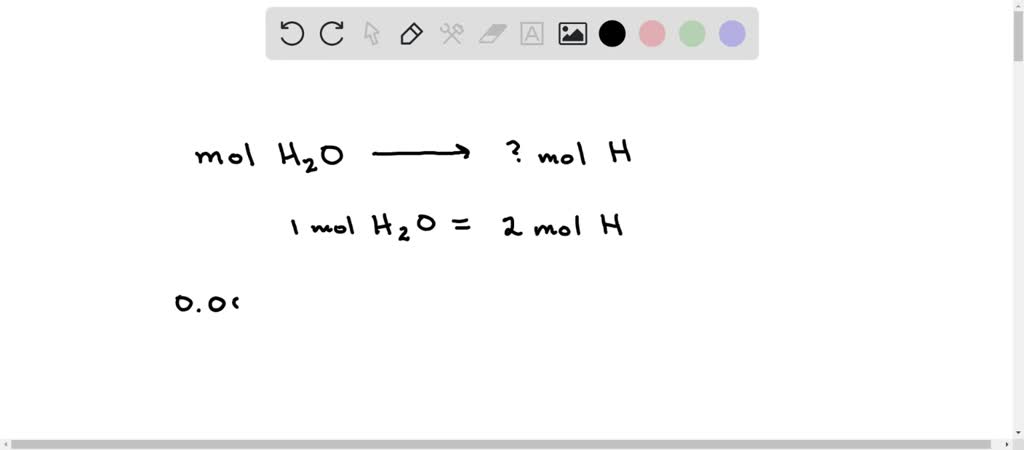
SOLVED: During the analysis, 0.00905 mol H2O is formed. Calculate the amount (mol) H in 0.00905 mol H2O.

The molar mass of H2O is 18.01 g/mol . The molar mass of 02 is 32.00 g/mol What is the mass of H20 in - Brainly.com
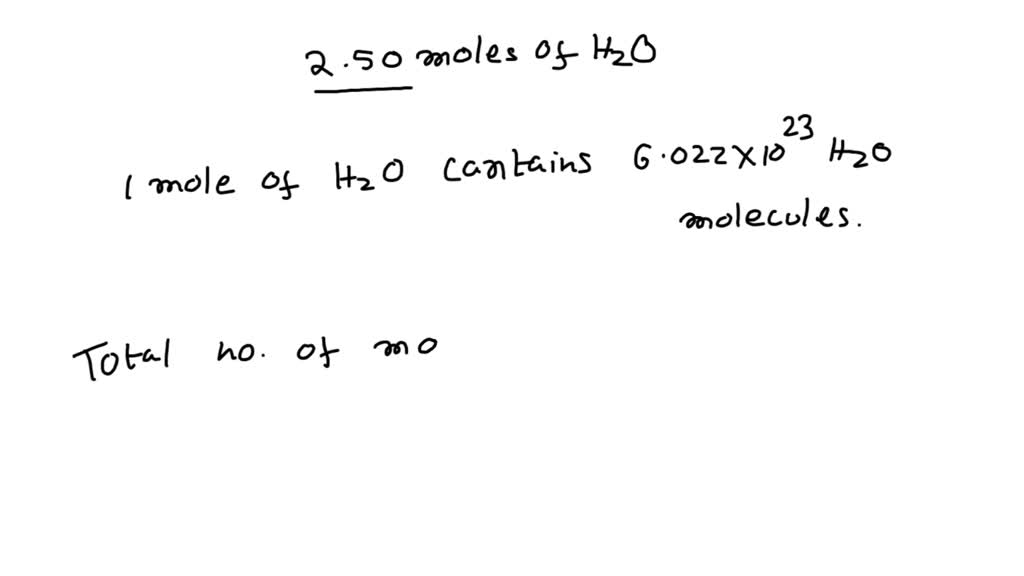
SOLVED: How many molecules are in 2.50 moles of H2O? 0 1.51 X 1024 molecules of H20 0 4.15 X 10-24 molecules of H20 0 3.01 X 1024 molecules of H20 O 8.31 X 10-24 molecules of H20

Question Video: Calculating the Mass of Water Produced Given the Masses of Oxygen and Hydrogen | Nagwa