H2 Evolution from H2O via O–H Oxidative Addition Across a 9,10-Diboraanthracene | Inorganic Chemistry | ChemRxiv | Cambridge Open Engage
Kp for the reaction CO2 + H2 =CO + H2O is found to be 16 at a given temperature. Originally equal number of moles of H2 and CO2 were placed in the

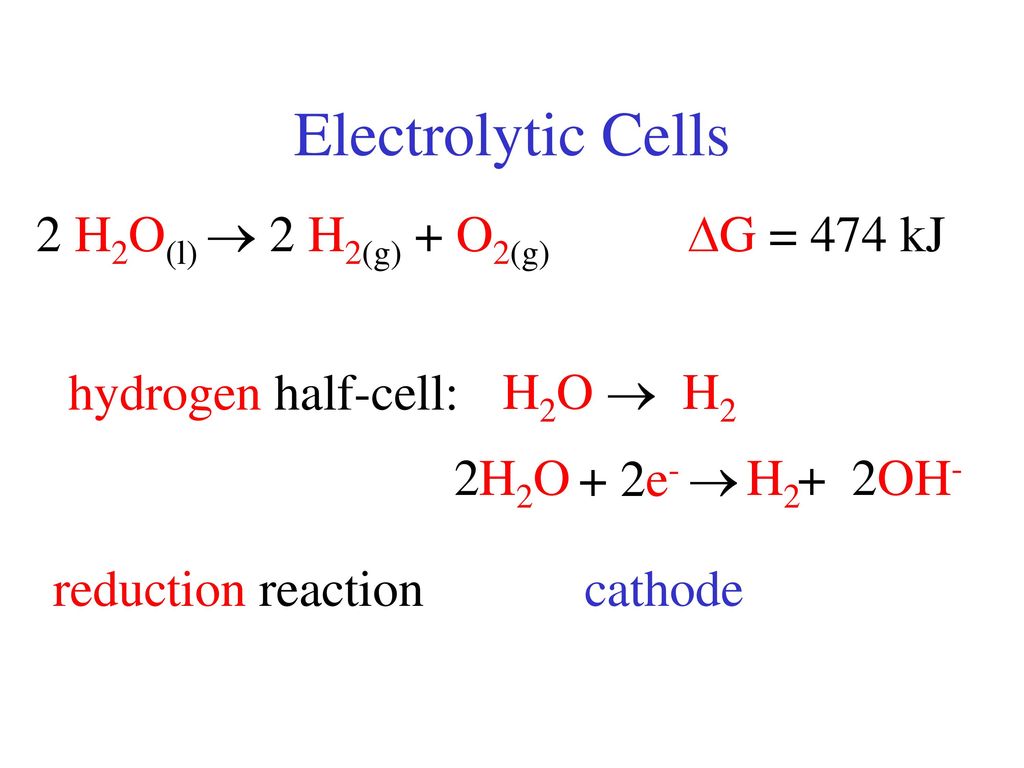
See: hydrogen reacts with oxygen to form water (H2O) according to the following equation: 2H2 + O2 → 2H2O - Brainly.com
Mixed H2O/H2 plasma-induced redox reactions of thin uranium oxide films under UHV conditions - Dalton Transactions (RSC Publishing)

calculate the equilibrium constant of H2 + O2 gives us H2O + CEO at 13957 if the equilibrium constant 135 - Chemistry - Equilibrium - 13886927 | Meritnation.com

n mole each of h2o h2 o2 r tken in closed container at temperature t if y mole of h2 r disasssociated at equillibrium n equillibrium pressure is p the cgt66gee -Chemistry -

Reaction of CO, H2O, H2 and CO2 on the clean as well as O, OH and H precovered Fe(100) and Fe(111) surfaces - Catalysis Science & Technology (RSC Publishing)
![SOLVED: The equilibrium constant expression Kc= [H2]^2 [O2]/[H2O]2 is the equilibrium constant expression for which of the following reactions? 2 H2 (g) + O2 (g) → 2 H2O (g) H2O (g) → SOLVED: The equilibrium constant expression Kc= [H2]^2 [O2]/[H2O]2 is the equilibrium constant expression for which of the following reactions? 2 H2 (g) + O2 (g) → 2 H2O (g) H2O (g) →](https://cdn.numerade.com/ask_previews/3f5fd1c4-1866-4d28-a520-21b96586832f_large.jpg)